 The total number of particles within the lattice can be calculated as follows: 8 particles on the lattice corners (1 particle),6 particles with half of their volumes within the lattice (6 particles* 1/2 volume = 3 particles). The two-dimensional layer representation shows that there are six particles which have half of their volumes within the lattice. It has a particle in the middle of each of the six faces of the cube. Illustrated left is the face-centered cubic (fcc) unit cell. The virus that causesfoot and mouth disease in animals also exhibits bcc crystal arrangements. Potassium (alkali metal) and iron are examples of metals that arrange themselves in bcc. The ratio of the volumes where all particlesare equal gives us a 68.0%. But we should realize that the lattice volume has increased accordingly. We might expect to have a greater packaging efficiency because there are more particles within the lattice compared to a simple cubic cell. The number of particles in a bcc unit cell is 2, determined as follows: 8 spheres on the lattice corners each with an eighth oftheir volume within the cell (1 particle) and one sphere completely embedded in the lattice. To determine the edge value we must rely on the diagonal of the two opposite corners of the unit cell. 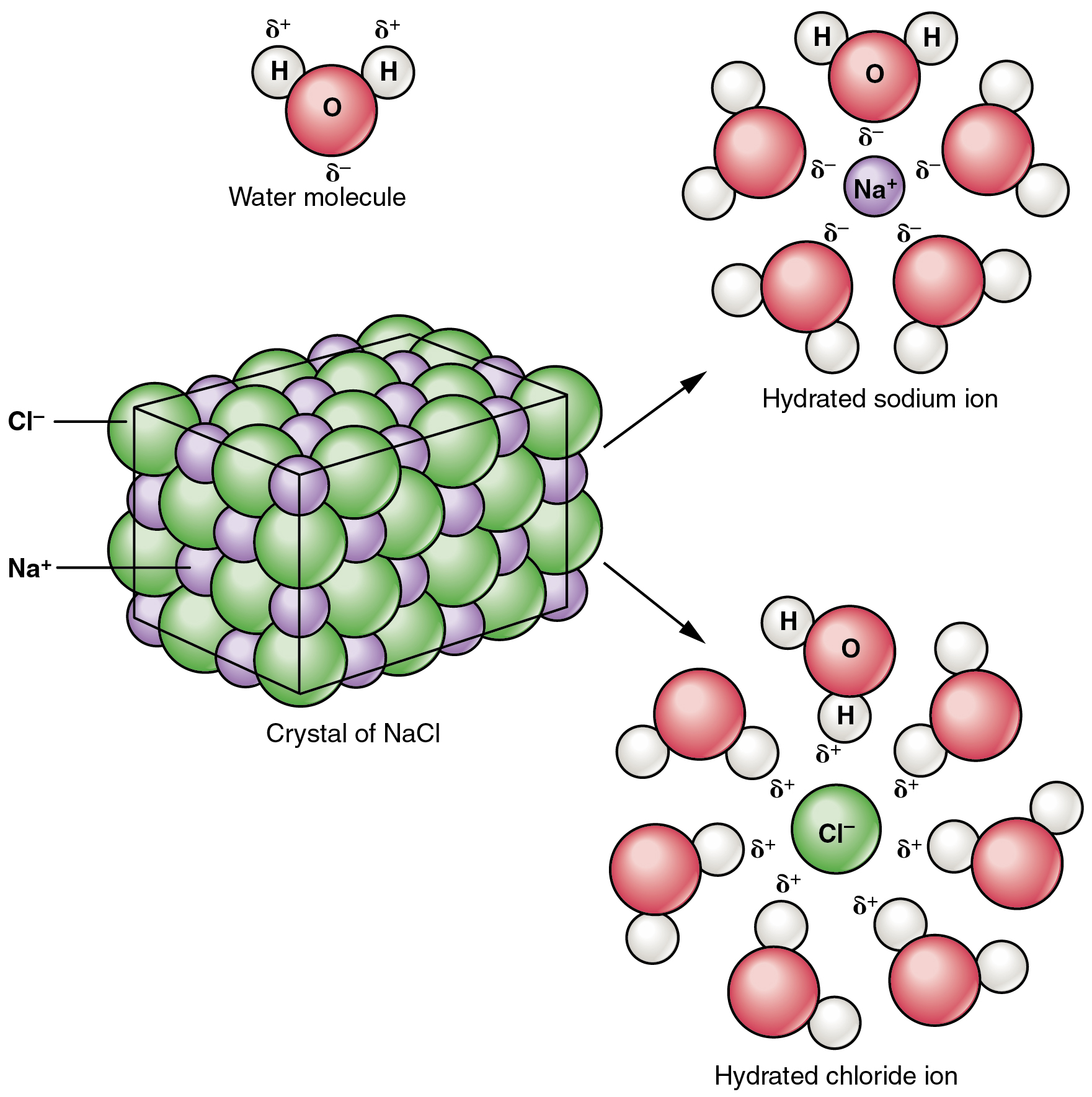
So in this case the edge is greater than two atomic radii of the lattice corners. The bcc has 3 layers (along the z-axis),which would look like this: Another way of looking at the layout is using 2-dimensional layer diagrams. As the name suggests it contains an ion or atom in the center of the cube.If all the spheres have the same radius, like in metals, then the spheres centered on the lattice points do not make contact with each other. Rotate the body-centered cubic (bcc) unit cell. One important question should be asked: what is the efficiency of packing same size atoms in simple cubes? 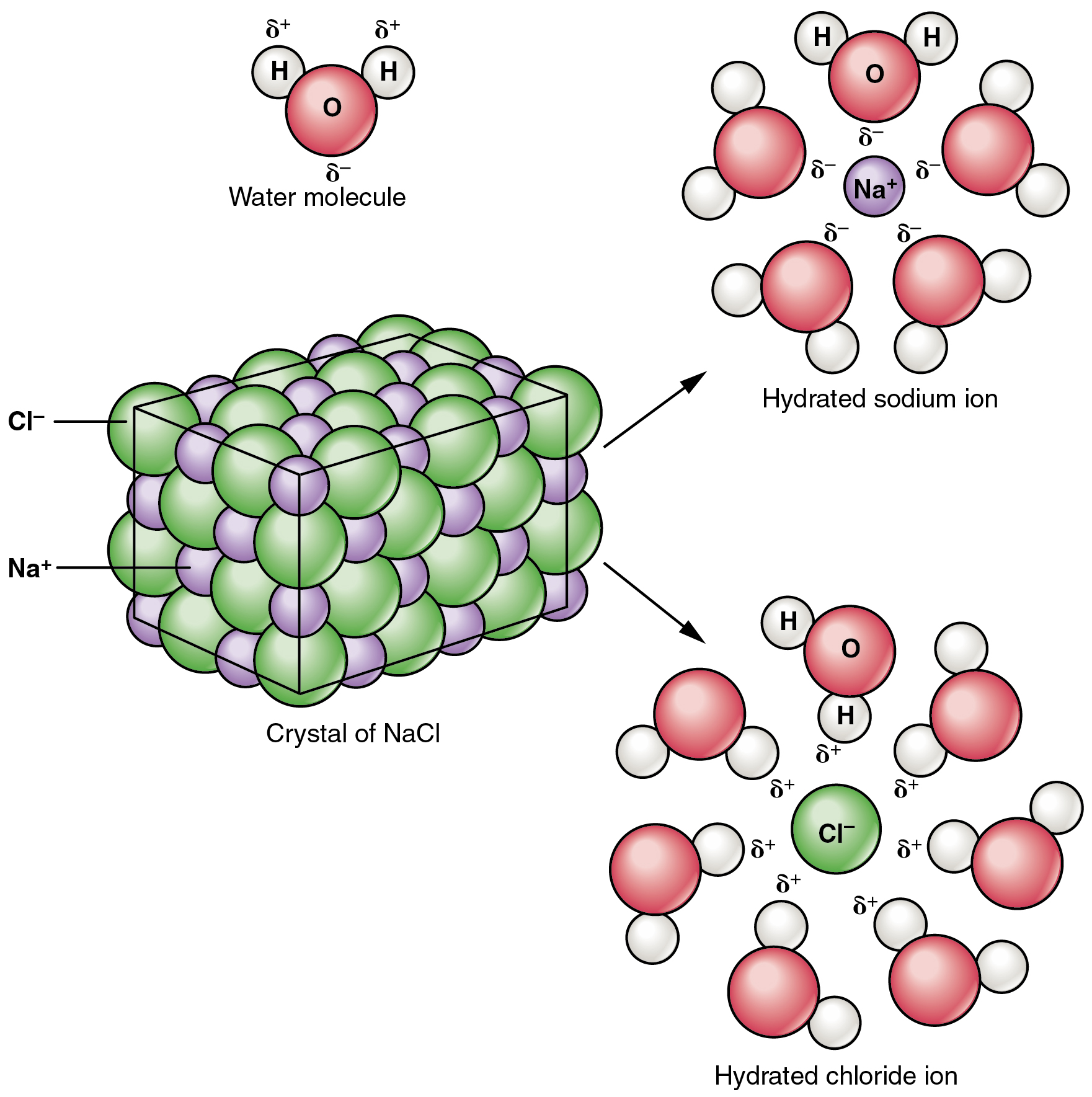 
Because there are a total of eight one eighth volume spheres in the cell the simple cubic unit contains one net particle. But the unit cell only contains, on the lattice points, an eighth of the volume of the sphere (ion or atoms). The volume of the unit cell then is the edge cubed (edge 3). Observe that in the simple cubic cell the edge equals two atomic radii. Return to the giant ionic structures page.As you rotate the spacefill model around you will notice that all the spheres (ions or atoms) are in contact with each other. If you find two chloride or two sodium ions with a direct line joining them, you have done it wrong! When you have finished, always check that every lattice line has a sodium ion at one end and a chloride ion at the other. That will force you to think rather than just copy. To be sure you know exactly what you are doing, it would probably be a good idea to draw this twice - once with the chloride ions on the corners of the big cube, and once with sodium ions on those corners. All that matters is that you alternate the ions so that you never get two ions the same joined directly by one of the lattice lines. In the top layer of ions, it doesn't matter whether you start with sodium ions or chloride ions in the corners. The exact numbers don't matter - just show the chloride ions as bigger. All the flaws will get covered up when you add the ions.ĭon't forget to add a key showing which ion is which.Ĭhloride ions have a diameter which is roughly twice the size of the sodium ions. The sketches above are definitely flawed! We are doing chemistry, not technical drawing. When you have done each face, it will look like this:įinally, you have to draw three lines through the centre of the cube from the centre of each face to the centre of the opposite face.ĭon't worry if, when you have finished, the lines don't meet exactly, or it looks a bit tatty. Start by drawing lines from the mid-points of each edge to the edge opposite on the same face of the cube. Now join the two squares together to make a cube. If you take the diagram above as a guide, you won't go far wrong. If you get it wrong, when you draw the ions, they will all end up in a muddle! The only way you are going to find this out is by trial and error. Then draw an identical square, offset from the first one. It simply describes how to draw the structure of a sodium chloride lattice This page is an add-on to a page about giant ionic structures. How to draw a simple sodium chloride lattice 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
